Development of Advanced Neural Decoding Algorithms for Next Generation Neuroprosthetics
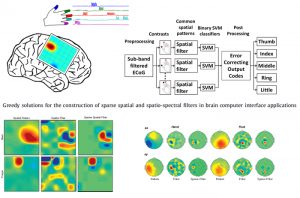
The objective is to develop novel feature extraction and signal processing algorithms to decode the neural activity of brain for a neuroprosthetics. In particular, we utilize adaptive signal processing and compressive sensing techniques to extract parsimonious features from high-density neural recordings from non-human primates [Ince et al., PlosONE 2010] and as well as human subjects [Onaran et al., BSPC 2012]. We collaborate with several institutions in the US in this particular direction.
Neuromarker Discovery for the Optimization of DBS
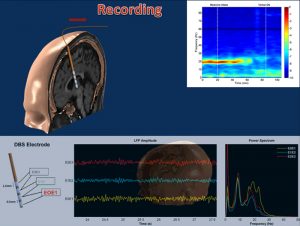
The goal of this work is to explore the neural activity of the brain to identify neurobiological markers for the optimization of deep brain stimulation in Parkinson Disease. We collaborate with Dr. Aviva Abosch, in this particular direction and obtain data from the sub thalamic nucleus of Parkinsonian subjects who are implanted DBS electrodes. Recently, we investigated the relationship between local field potential (LFP) activities in the subthalamic nucleus and the therapeutic response to programming. We showed that postoperative subband analysis of LFP recordings in β and γ frequency ranges may be used to select optimal electrode contacts. These results indicate that LFP recordings from implanted DBS electrodes can provide important clues to guide the optimization of DBS therapy in individual patients [Ince et. al, Neurosurgery 2010].
Investigation of Neural Biomarker in Epilepsy using Computational Intelligence
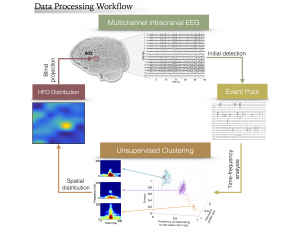 This project aims at the investigation of neural-biomarkers in patients with refractory epilepsy, for the purpose of accurate seizure onset zone (SOZ) identification and assisting in surgical planning. Epilepsy is one of the most common neurological diseases. Surgical resection of SOZ can provide seizure freedom for many patients with drug resistant epilepsy, which requires invasive EEG (iEEG) monitoring over an extended period of time and detailed visual inspection of collected data by neurologists. High frequency oscillations (HFO, 80 – 500 Hz) are highly valued as a promising clinical biomarker for epilepsy. Due to the low amplitude and short duration of these events, visual identification of HFO transients in the clinical recordings of vast size is infeasible, which has limited the utilization of HFOs as SOZ indicators in clinical practice. In the current pr
This project aims at the investigation of neural-biomarkers in patients with refractory epilepsy, for the purpose of accurate seizure onset zone (SOZ) identification and assisting in surgical planning. Epilepsy is one of the most common neurological diseases. Surgical resection of SOZ can provide seizure freedom for many patients with drug resistant epilepsy, which requires invasive EEG (iEEG) monitoring over an extended period of time and detailed visual inspection of collected data by neurologists. High frequency oscillations (HFO, 80 – 500 Hz) are highly valued as a promising clinical biomarker for epilepsy. Due to the low amplitude and short duration of these events, visual identification of HFO transients in the clinical recordings of vast size is infeasible, which has limited the utilization of HFOs as SOZ indicators in clinical practice. In the current pr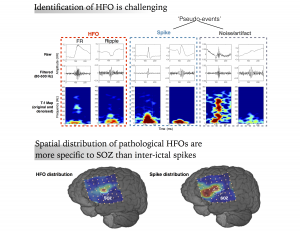 oject, we mainly focus on the quantitative detection and characterization of HFOs in continuous human iEEG data using machine learning and other computational methods. We develop novel algorithms to effectively detect HFO and its subtypes in massive clinical iEEG datasets in an automated fashion, refine the detection by isolating HFOs from inter-ictal spikes and other non-neural events, and investigate the prognostic value of signature activities such as HFOs and interictal spikes.
oject, we mainly focus on the quantitative detection and characterization of HFOs in continuous human iEEG data using machine learning and other computational methods. We develop novel algorithms to effectively detect HFO and its subtypes in massive clinical iEEG datasets in an automated fashion, refine the detection by isolating HFOs from inter-ictal spikes and other non-neural events, and investigate the prognostic value of signature activities such as HFOs and interictal spikes.
Intraoperative Localization of STN in Parkinson’s Disease by using Microelectrode LFPs
 Deep brain stimulation (DBS) of the subthalamic nucleus (STN) is an effective and the commonly used surgical treatment for the motor symptoms of Parkinson’s disease (PD). DBS involves the surgical implantation of a quadripolar electrode into the motor territory of STN, followed by connection to a programmable hardware (implanted pulse generator; IPG) in the patient’s chest. The IPG is subsequently programmed to deliver stimulation to the STN. However, stimulation can result in side effects arising from the spread of stimulation to structures surrounding the STN. Thus, the clinical efficacy of DBS depends critically on accurate localization of the STN.
Deep brain stimulation (DBS) of the subthalamic nucleus (STN) is an effective and the commonly used surgical treatment for the motor symptoms of Parkinson’s disease (PD). DBS involves the surgical implantation of a quadripolar electrode into the motor territory of STN, followed by connection to a programmable hardware (implanted pulse generator; IPG) in the patient’s chest. The IPG is subsequently programmed to deliver stimulation to the STN. However, stimulation can result in side effects arising from the spread of stimulation to structures surrounding the STN. Thus, the clinical efficacy of DBS depends critically on accurate localization of the STN.
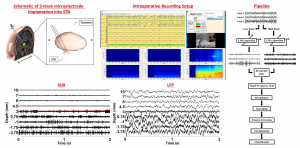 In practice, targeting of the STN during DBS surgery with imaging modalities is followed by intraoperative electrophysiological techniques consisting of the conversion of neural activity, in the form of single-unit neuronal activity (SUA) recorded at the microelectrode tip, into audio and visual signals. However, MER/SUA method is qualitative, subjective, and has certain limitations. Local field potentials (LFPs) represent the activity of a population of neurons and excessive beta band (11-30Hz) LFP activity is frequently reported in STN. In this regard, for the purpose of assisting with clinical decision making and increase the accuracy of implantation of the electrode into the motor territory of STN, we developed an automated approach for electrophysiological localization of STN. We suggested that fused with existing mapping techniques, using this technique online in the operating room may increase the accuracy of the DBS macroelectrode placement. In this way, it might contribute to the efficacy of DBS by reducing the stimulation voltage and associated side effects. Since the electrode placement is guided by LFP activity, the current technique could also be useful to monitor the LFP events which are capable to fine-tune the future DBS settings in a closed loop
In practice, targeting of the STN during DBS surgery with imaging modalities is followed by intraoperative electrophysiological techniques consisting of the conversion of neural activity, in the form of single-unit neuronal activity (SUA) recorded at the microelectrode tip, into audio and visual signals. However, MER/SUA method is qualitative, subjective, and has certain limitations. Local field potentials (LFPs) represent the activity of a population of neurons and excessive beta band (11-30Hz) LFP activity is frequently reported in STN. In this regard, for the purpose of assisting with clinical decision making and increase the accuracy of implantation of the electrode into the motor territory of STN, we developed an automated approach for electrophysiological localization of STN. We suggested that fused with existing mapping techniques, using this technique online in the operating room may increase the accuracy of the DBS macroelectrode placement. In this way, it might contribute to the efficacy of DBS by reducing the stimulation voltage and associated side effects. Since the electrode placement is guided by LFP activity, the current technique could also be useful to monitor the LFP events which are capable to fine-tune the future DBS settings in a closed loop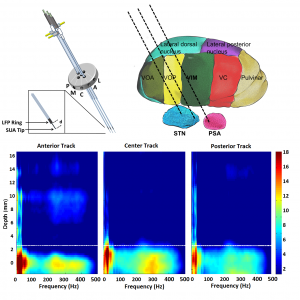 paradigm.
paradigm.
In addition to the practical advantages, the current approach also provided an insight into the neurophysiology of PD by describing the spatio-spectral LFP patterns. Depth-frequency maps (DFM) providing a concise summary of the depth varying frequency content of LFPs indicated that beta oscillations are getting stronger as the electrodes approach the STN. Not only beta oscillations but also strong high frequency oscillations (HFOs) can be observed in the STN area well aligned with beta oscillations.
